The Xu Lab: The Distribution of Infectious Disease and Variance in Ecological Pressures Greatly Influences Fungal Evolution
Dr. Jianping Xu is an active member of the Michael G. DeGroote Institute for Infectious Disease Research (IIDR) and David Braley Centre for Antibiotic Discovery (DBCAD) who studies fungal biology. In 2000, Dr. Xu joined McMaster University where he began working as an assistant professor in the Department of Biology after completing his post-doctorate at Duke University. Now, he is a tenured full professor who enjoys teaching undergraduate, graduate, and post-graduate students. Dr. Xu, along with his team of dedicated researchers, aims to advance our understanding of fungal pathogenesis and the global health impact of microbial evolution. The Xu lab tackles new avenues for infectious disease research and continues to expedite scientific breakthroughs in the field of ecology and evolutionary genetics of microorganisms at McMaster University.
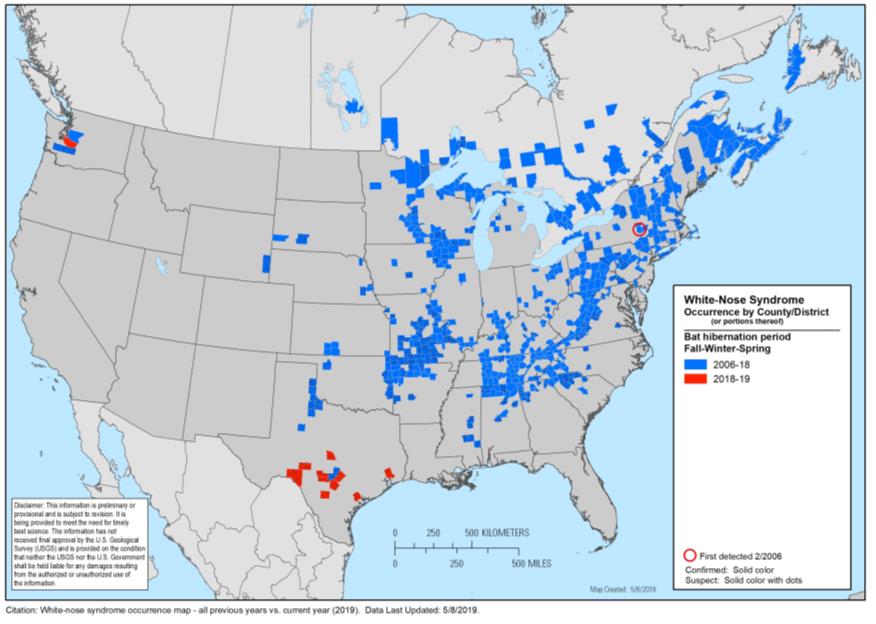
Dr. Xu’s interest in population genetics (one of many) often brings him to unexpected places. One of his most recent projects, featured in several social media networks, involves Pseudogymnoascus destructans (Pd) – a fungal pathogen that causes white-nose syndrome (WNS) and widespread mortality amongst vulnerable hibernating little brown bats (Myotis lucifugus). WNS first made its appearance in New York State in 2006. More than a decade later, the occurrence of WNS expanded to approximately 33 American states and 5 eastern Canadian provinces, contributing to over 7 million deaths. As of 2019, WNS is predicted to threaten western NA, particularly the caves in Vancouver.
The project was first established in 2014 by Masters student Jordan Khanket along with Dr. Xu. Fungal isolates collected from 2008 to 2013 were provided by the University of Guelph, New Brunswick Museum, the Canadian Cooperative Wildlife Health Center, and the Mycology laboratory of the New York State Public Health and screened for potential phenotypic variations to multiple environmental variables. Their results, published in PLOS ONE in 2014, found that clonal expansion is accompanied by significant phenotypic variation but limited genotypic variation. They suggest that this may occur due to the rapid accumulation of spontaneous genetic mutations; however, the exact genetic basis requires further investigation.
Recently, Adrian Forsythe, a PhD student in Dr. Xu’s lab, revealed that the WNS pathogen may have evolved to adapt to different ecological niches along geographic gradients across NA. Their recent work looks at geographic variation specifically climate differences that can produce a temperature gradient to support the rapid phenotypic diversification and clonal expansion of Pd. Hibernation is a critical time point for bats to remain in deep slumber. Once exposed to WNS, bats wake up and starve due to energy loss, and exhaustion.
As we edge towards regional extinction of Myotis lucifugus, the search for a cure has become a top priority for the Xu lab. They believe that understanding genotypic and phenotypic variation of pathogens can help manage the spread of infectious diseases. Their research brings to light the consequences of persistent infections in natural environments. Bats are experts in plant pollination and are crucial for regulating mosquito populations. Today, Dr. Xu works closely with Dr. Naowarat Cheeptham from Thompson Rivers University in British Columbia and Dr. Cori Lausen of Wildlife Conservation Society (WCS) Canada to develop a probiotic “cocktail” that can inhibit Pd growth in an effort to control WNS.
Beyond bats, Dr. Xu is also interested in fungal diseases in humans. They use microbiological, molecular, ecological and quantitative genetic tools to answer new and exciting research inquiries. As a whole, the lab works on diverse fungal species and collaborates broadly with researchers from other countries.
The Xu’s lab outstanding contributions to the scientific community was recognized by the Genetics Society of Canada and the Canadian Botanical Association. Dr. Xu sits on various editorial boards and is the recent recipient of the development of biomarkers for efficient identification of human fungal pathogens grant. Outside of science, he competes in high-profile race walks and holds several national records. To learn more about the Xu lab’s impactful research, visit their website at www.xulabmcmaster.com.
NewsRelated News
News Listing

Brighter World ➚
Researchers invent artificial intelligence model to design new superbug-fighting antibiotics
News
March 23, 2024
FHS News ➚
Decades of McMaster tuberculosis research leading to new prevention and control strategies
News
March 22, 2024


